Dishwashing soaps and gels significantly speed up the process of cleaning dishes and other utensils. Nevertheless, you need to choose the right household chemicals. Some dish soaps have an aggressive composition.
They can cause allergies and other adverse reactions that have a bad effect on the human body. So, the use of any household chemicals should begin with an examination of the information on the label.
Dish Soap: pH 3 to 11.5
PH neutral dish soaps (5.5-8) coincide with the pH of our skin and are harmless to the skin of the hands. You should know that pH of ordinary dish soap may go beyond a neutral pH level. So it is perfect if you use gloves when washing dishes.
Simple Explanation of PH
We often hear about the pH level of various substances in advertising, in programs about food and health, we see this information on the goods labels. And each manufacturer is trying to prove to us that their pH is the best. Let’s see what mysterious letters p and H mean. It’s simple!
- p is Lat. pondus (weight), in other words, it means “indicator”
- H is a chemical element called “hydrogen”
- Hence, pH is the “hydrogen indicator”.
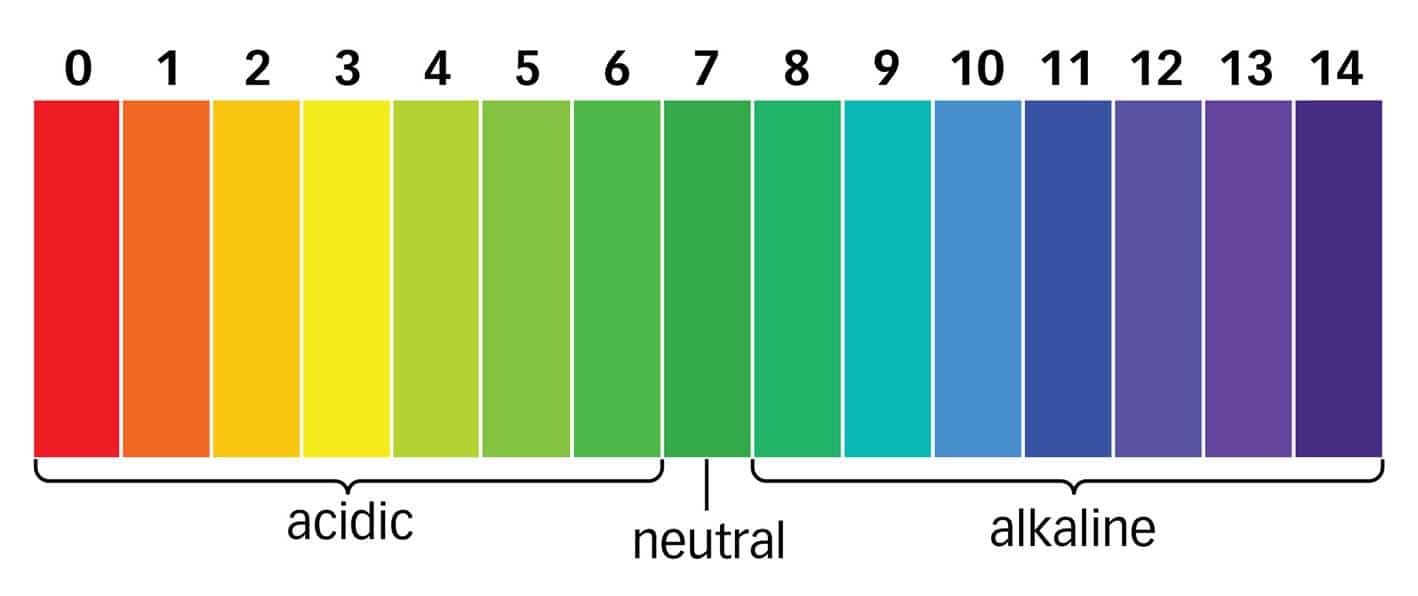
All substances have either an acidic or alkaline environment.
What Is Neutral PH Level?
You also must have heard about a neutral environment. So, pure water is a clear example of a neutral environment. This parameter equals 7. And we use a scale from 0 to 14 to determine the pH level.
- From 0 to 6.5 is the acidic environment. The lower the indicator, the more acidic the substance is.
- From 6.5 to 7.5 is a neutral pH level.
- From 7.5 to 14 – alkaline environment.
PH of Detergents
Soaps, shower gels, and other detergents are made on the basis of alkalis. For example, the hand soap pH is 9.0-10.0. Detergent manufacturers are trying to reduce the pH even not to a neutral environment (6.5-7.5), but to the pH of human skin.
[expert_review_poll id=”6743″ params=”JTdCJTIydGl0bGUlMjIlM0ElMjJXaGF0JTIwZG8lMjB5b3UlMjB1c2UlMjBmb3IlMjB3YXNoaW5nJTIwZGlzaGVzJTNGJTIyJTJDJTIyc2hvd190aXRsZSUyMiUzQSUyMjElMjIlMkMlMjJzaG93X2NvdW50JTIyJTNBJTIyMCUyMiUyQyUyMnN0eWxlJTIyJTNBJTIybGlnaHQtMSUyMiUyQyUyMmNvbG9yJTIyJTNBJTIyYmx1ZS0xJTIyJTJDJTIybXVsdGlwbGUlMjIlM0FmYWxzZSUyQyUyMmFuc3dlcnMlMjIlM0ElNUIlN0IlMjJpZCUyMiUzQSUyMjElMjIlMkMlMjJ0ZXh0JTIyJTNBJTIyRGlzaHdhc2hpbmclMjBEZXRlcmdlbnRzJTIyJTdEJTJDJTdCJTIyaWQlMjIlM0ElMjIyJTIyJTJDJTIydGV4dCUyMiUzQSUyMk5ldXRyYWwlMjBkZXRlcmdlbnRzJTIyJTdEJTJDJTdCJTIyaWQlMjIlM0ElMjIzJTIyJTJDJTIydGV4dCUyMiUzQSUyMkJvdGglMjIlN0QlMkMlN0IlMjJpZCUyMiUzQSUyMjQlMjIlMkMlMjJ0ZXh0JTIyJTNBJTIyTmVpdGhlciUyMG9uZSUyMG5vciUyMHRoZSUyMG90aGVyJTIyJTdEJTVEJTdE”]
Otherwise, the skin dries up, it becomes allergic and itchy. To restore balance, the body spends a lot of water and microelements.
What is the PH of Dishsoap?
Dish soap pH levels can vary from 3 to 11.5. The pH indicator tells about the acidity of the solution. PH neutral soaps (5.5-8) coincide with the pH of our skin and are harmless to the skin of the hands.
However, it is assumed that when washing dishes, it is better for people to protect their hands with gloves, therefore standards for pH of detergents may go beyond neutral the pH level.
What Is a Neutral PH Cleaner?
Neutral detergents are applied manually or using auxiliary technical me
ans for applying to fresh contaminants. Neutral detergents leave no residue and remove external impurities while giving the surface shine.

The main advantages of neutral pH cleaners:
[expert_review_poll id=”6744″ params=”JTdCJTIydGl0bGUlMjIlM0ElMjJEbyUyMHlvdSUyMHVzZSUyME5ldXRyYWwlMjBQSCUyMENsZWFuZXJzJTNGJTIyJTJDJTIyc2hvd190aXRsZSUyMiUzQSUyMjElMjIlMkMlMjJzaG93X2NvdW50JTIyJTNBJTIyMCUyMiUyQyUyMnN0eWxlJTIyJTNBJTIybGlnaHQtMSUyMiUyQyUyMmNvbG9yJTIyJTNBJTIyYmx1ZS0xJTIyJTJDJTIybXVsdGlwbGUlMjIlM0FmYWxzZSUyQyUyMmFuc3dlcnMlMjIlM0ElNUIlN0IlMjJpZCUyMiUzQSUyMjElMjIlMkMlMjJ0ZXh0JTIyJTNBJTIyWWVzJTJDJTIwb2Z0ZW4lMjIlN0QlMkMlN0IlMjJpZCUyMiUzQSUyMjIlMjIlMkMlMjJ0ZXh0JTIyJTNBJTIyWWVzJTJDJTIwYnV0JTIwcmFyZWx5JTIyJTdEJTJDJTdCJTIyaWQlMjIlM0ElMjIzJTIyJTJDJTIydGV4dCUyMiUzQSUyMk5vJTJDJTIwbmV2ZXIlMjIlN0QlNUQlN0Q=”]
- environmental safety, biodegradability (without emission of harmful substances);
- ability to regenerate (after sedimentation it can be used repeatedly, up to 2-5 times);
- fire and explosion safety;
- solubility and effectiveness in the water of any hardness;
- lack of phosphates.

Benefits of Neutral pH Cleaners
- Environmental Safety: These cleaners are biodegradable and do not emit harmful substances.
- Reusability: They can be regenerated after sedimentation and reused multiple times (up to 2–5 times).
- Safety: They are fire and explosion safe.
- Versatility: Effective in water of any hardness and free from harmful phosphates.
PH Level of Laundry Detergent
There are strict regulations regarding the most dangerous substances that are part of detergents, namely phosphates, surfactants, and pH levels of laundry detergents. A high concentration of acid or alkali is dangerous not only for the skin but also for the clothes.
So, phosphates should not exceed 17%, surfactants should be biodegradable for at least 80%.
And with regard to pH, this indicator is adjusted depending on which types of fabric the detergent is meant.
The concentration level of hydrogen ions should be within the range from 7.5 to 11.5. The closer the indicator is to the lower border, the better it is for the clothes and linen as they wear less during washing.

The PH of Various Household Cleaners
To help keep your head from assumptions about how the pH scale is used in cleaning, let’s see what pH level traditional cleaning products have.
Chlorine bleach (pH 11 – 13)
Is an extremely alkaline product, which makes it fantastic for whitening and stains removing. The position of the bleach pH on the pH scale indicates its corrosion. This means it is great for any solid surface, but you need to have adequate ventilation when using it.
Toilet Cleaner (pH 1 – 3)
On the opposite end of the scale, we have toilet cleaners, which are extremely acidic but perfect for destroying minerals and other inorganic abominations that may accumulate in your toilet. Since the product is very acidic, you should use these products with extreme caution.
Soft Dishwashing Liquids (pH 7 – 8)
If you use dishwashing detergents marked as soft or pH neutral soaps, it usually means that their pH is just in the middle. And it makes them perfect for daily cleaning. Soft agents will not damage most surfaces, so you may not use any protection when handling neutral acid products.
Ammonia (pH 11 – 12)
It is another very alkaline substance. Ammonia is a formidable detergent that can easily cope with removing dirt and soot. Its position on the scale means that it must be used with adequate protective equipment in areas with sufficient ventilation.
Vinegar (pH 3)
This cleaning agent is on the opposite end of the pH scale. The acidic properties of vinegar make it ideal for the removal of entrenched mineral deposits, but at the same time, it is a poor choice for some surfaces (in particular, for stone).
Vinegar can also cause real harm if you do not take precautionary measures. So protection of face and hands is a must.

What components should neutral cleaning products contain?
- Soda, it is food reagent, softener and pH regulator;
- Sodium citrate or citric acid salt. It is a preserving agent that has an antibacterial substance. It creates a foam and regulates the pH level;
- oxygen bleach, it fights stains;
- enzymes, they fight against protein and fat pollution;
- essential oils if you do not have individual intolerance.
How to Test the pH of Your Dish Soap at Home
Your dish soap’s pH level is crucial since it affects skin safety as well as cleansing power. Testing the pH at home requires only a few instruments and simple ingredients.
Materials Needed
- pH test strips: they can be purchased online or at pharmacies and pool supply stores.
- Distilled water: it removes contaminants, thereby guaranteeing reliable results.
- Clean container: To combine the solution of soap.
- Measuring tools: To guarantee accurate ratios of dilution.
Procedure
- Create the Soap Solution: In the clean container, measure one teaspoon of your dish soap then add it to one cup of distilled water. Stir until the soap dissolves entirely.
- Dip the pH Test Strip: Submerge a pH test strip in the soap solution for the time duration indicated on the strip’s label, usually a couple of seconds.
- Review the findings: After removing the strip, match its color change to the given pH scale chart. This will show the pH of your dish soap.
Interpreting the Results
- pH 7: (neutral) appropriate for daily usage; mild on skin.
- pH less than 7: (acidic) could be harmful for skin; think about wearing gloves.
- pH above 7: (alkaline) Good for removing grease, but may cause skin irritation; gloves are advised.
Testing the pH of your dish soap on a regular basis guarantees that it fits your cleaning requirements and skin sensitivity, therefore encouraging both good dishwashing and personal comfort.
What chemicals to avoid in dishwashing detergents?
When choosing a pH neutral cleaner, you should not forget to check it for the absence of harmful components.
- Phosphates/phosphonates. They have been banned in many countries. These components adversely affect the human body and cause irreparable damage to the environment.
- Synthetic phthalates / flavors. They can lead to endocrine disorders.
- Anionic surfactants. It will be very difficult to wash off the detergent if this component is available in its composition.
- Optical bleach/chlorine. These components lead to heavy allergies.
- Synthetic preserving agents. They are dangerous as they are not excreted in the body, moreover, they adversely affect the endocrine system.
It is best to choose eco-products that are hypoallergenic and biodegradable.
Mild Detergent For Any Purpose. How to Make It Yourself
Whenever we are headed to clean something in our kitchen or bathroom, we go for a bottle of the cleaning agent.
Since most of them contain harmful chemical ingredients that make those products harsh to our skin, people tend to ask more and more often whether there is a way to prepare a harmless solution to clean the house using simple and easy to get ingredients.
Fortunately, making such natural mild cleaning solutions is not a problem at all. You will only need some common ingredients that everyone has in their kitchen, and a bit of time to get the product ready. No special tools or skills are needed – isn’t that awesome?!
Another benefit you’ll get is that this cleaning product can be applied in any area from the kitchen to the bathroom so be ready that you may need a bit more than one bottle!
As a result, such a detergent will take care not only of your household but also of yourself.
Natural dish detergent
For this purpose, you will need a bottle of pH-neutral dishwashing liquid and hot water.
Take a quarter cup of the agent and mix it in a gallon of water in a bucket.
Done! It will work the same well and will clean the dishes effectively only without harming your skin.

Go for distilled vinegar
Another option of preparing an all-clean product yourself is to search for white vinegar on your kitchen shelves. This natural liquid will work both to clean the surfaces and deodorize/disinfect them.
The ingredients are super simple: one cup vinegar and a gallon of hot water. Mix together and go ahead! This natural solution can clean anything being a great all-purpose product.
Try mustard to clean the dishes
Only not the one that’s sold in bottles.
We mean powdered yellow mustard since it can work the same well as any chemical product!
No additional ingredients, just a packet of this yellow powder – and your mugs and tableware will be shiny again. Besides, mustard is well known for its gentle care about our skin.
All benefits, no harm!
After preparing a homemade cleaning solution, put it in a bottle, sign it so that you could know what’s in there, and keep it by hand in case you need to clean something.
Such natural cleaning products require very little effort to make them and they care about our skin and house way better than the chemical ones being much safer in addition.
Frequently Asked Questions
Dish soap is a frequently used washing product in any home. However, is it so safe or not? Shall we protect our hands from it? Find out the answers below.
What Is pH of a Dish Soap?
Many of us don’t know what those pH letters mean. In fact, it is the measurement of the hydrogen ions concentration comparing to the concentration of hydroxyl ions. If there is an excess of hydrogen over hydroxyl. we get acid. If vise versa, it is called basic (or alkaline).
In its turn, all the products made of water have their pH with its lowest rate of 1 and the highest rate of 14. Products whose pH is from zero to 6.9 refer to acids, whilst the pH number from 7.1 to 14 shows that we deal with a basic. Accordingly, the pH rate of 7 is neutral.
What Is pH of Dawn Dish Soap?
Dawn is a famous label that produces cleaning products. According to the Materials Safety Data Sheets, the pH level of Dawn dish soap is between 8.7 and 9.3 which makes this washing product moderately basic. In its turn, it means that you don’t need any protection like gloves when you wash dishes with it.
What Is the pH Level of Dish Soap?
For a dish soap, the pH level must be above 7 (9 or 10 is best) as then the product will be more effective to grease. Products with a lower pH level will have less effect and will be more harmful to your skin.

I know Ph 7-8 is best but you tell 9-10 is best. What Ph is best in liquid dishwash?
The Materials Safety Data Sheet (MSDS) for Dawn dish detergent states (you can google it, cant put a link here) that Dawn has a pH of 8.7 – 9.3. That means it’s moderately basic. So it is a standard, and you can use it without any harm.
Cleaning criteria do not decide by pH , it is decided by ingredients used in it . For dish wash recommended, pH is 8 to 10. Because 7 is neutral, and you will need much more effort to deal with the stains.
Because after 10 ph goes chlorine pH 11 to 13, ammonia: pH 11 to 12 and so on.
So you can choose any brand that you prefer, and for the most greasy part of the kitchen, choose smth special with 9 to 10 ph.
What is the pH of coffee? I heard many times that people use it as a skincare product or a scrub. Is that safe doing this?
Well, if it is a brewed coffee, its pH level is about 4.8 to 5.10 which makes it acidic. However, when applied externally, this product can, on the contrary, fight some skin problems like acne. I also heard it has anti-aging effect but I didn’t have an occasion to check!
Does dishwashing liquid kill bacteria? I’m thinking about shifting to non-chemical means so the question is pretty crucial. Thanks!
Yes, it does kill germs and bacteria just like hand soap does.
But what’s the problem? Organic means are the same efficient being way less harmful to the skin and surroundings. I often use non-chemical soaps and other cleaning agents and I see no difference except for my skin stopped being irritated.
Any dishwashing liquid makes my skin itch and turn red. So now I’m thinking about trying something less traumatic. Which dish soap is pH neutral? Could you suggest anything?
Search for something with a pH of 7, it’s neither acidic nor alkaline. I know that Seventh Generation is good, also Puracy, Ecos, and Palmolive are fine. A friend of mine uses Frosch and sometimes Mrs. Mayer’s, she says both are well-working.
Is Dawn dish soap acidic or basic?
None of them. Its pH is 7.4 so I guess it is neutral.
Is Palmolive dish soap septic safe? I use it all the time since it is good for my skin, but what about the septic system?
It depends on which product exactly you mean since Palmolive has plenty of kinds of soaps and other household cleaning stuff. I know for sure that Palmolive Eco Detergent is safe for the septic tank, but I can’t be sure about the other products of this brand.
In our family, my husband never rinses plates after washing them with dish soap. Is dish soap residue dangerous? I always thought it must be washed away.
It can be harmful because any chemicals cause nausea and vomiting when we swallow them. So I guess that constant “eating” of residue (since it remains on the tableware) can one day result in an upset stomach. Anyway, if you have kids, your husband should be more careful since children have more sensitive stomachs.