Often, different things that sound obvious are interesting and even exciting. Sometimes such common information turns up quite useful!
Lots of people have definitely wondered at least once in their life what time it takes for water to freeze.
So, how long does it take for water to freeze? In a freezer, it will take from 1 hour to two hours if you what to get ice cubes at a temperature of 0° F. If your water is cold or really hot, the water will freeze even faster (around 45 minutes). This paradox we call the Mpemba effect.
It’s not a vitally important knowledge, however, in certain situations, it can be very handy (let’s say when we need to freeze something liquid for the party or just make some ice).
How fast can water freeze? Why not to search for a clue?
What Takes Place When Water Is Affected By Frost
We all have learned from school that the freezing point for water is zero degrees Celsius. To understand what processes take place when it starts freezing, let’s refresh some basic knowledge of physics.
It is known that water, just like any other liquid, consists of constantly moving molecules.
Whenever it is heated up, the molecules start moving much faster and when it cools down, they slow down their move respectively.
If during the heating the molecules can move so fast that they start to evaporate turning into a gas, freezing works on the contrary. When being exposed to frost, molecules become so slow that they start hooking to each other which leads to water hardening into a solid crystal.
As for water, unlike many other liquids, it expands in the hard condition and becomes denser than its liquid state.

How long does it take for the liquid to freeze?
It generally hangs upon the capacity. Anyway, normally, the approximate time is between three and four hours.
And now, let’s clarify how long it takes water of different heating degrees to freeze.
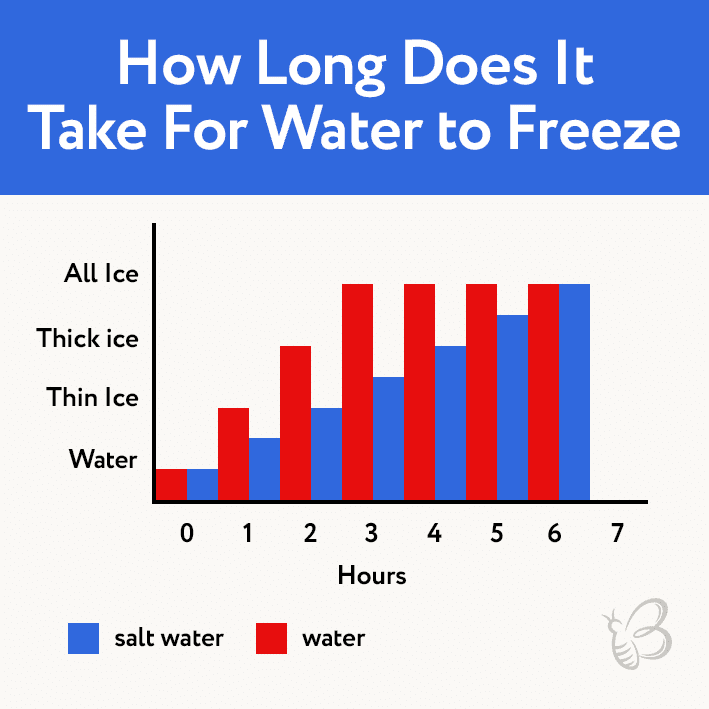
Does Water Of Different Heating Degrees Harden Equally Fast?

We are sure that many of you have noticed many times how water turns frozen quicker or slower hanging upon the temperature.
What is the reason for this?
[expert_review_poll id=”6938″ params=”JTdCJTIydGl0bGUlMjIlM0ElMjJIb3clMjBkbyUyMHlvdSUyMHVzdWFsbHklMjB1c2UlMjBpY2UlMjBjdWJlcyUzRiUyMiUyQyUyMnNob3dfdGl0bGUlMjIlM0ElMjIxJTIyJTJDJTIyc2hvd19jb3VudCUyMiUzQSUyMjAlMjIlMkMlMjJzdHlsZSUyMiUzQSUyMmxpZ2h0LTElMjIlMkMlMjJjb2xvciUyMiUzQSUyMmJsdWUtMSUyMiUyQyUyMm11bHRpcGxlJTIyJTNBZmFsc2UlMkMlMjJhbnN3ZXJzJTIyJTNBJTVCJTdCJTIyaWQlMjIlM0ElMjIxJTIyJTJDJTIydGV4dCUyMiUzQSUyMkklMjBhZGQlMjB0aGVtJTIwdG8lMjBjb2NrdGFpbHMlMjIlN0QlMkMlN0IlMjJpZCUyMiUzQSUyMjIlMjIlMkMlMjJ0ZXh0JTIyJTNBJTIySSUyMGFkZCUyMHRoZW0lMjB0byUyMHRlYSUyMiU3RCUyQyU3QiUyMmlkJTIyJTNBJTIyMyUyMiUyQyUyMnRleHQlMjIlM0ElMjJJJTIwYWRkJTIwdGhlbSUyMHRvJTIwdGhlJTIwanVpY2UlMjIlN0QlNUQlN0Q=”]
Let’s imagine that we have two tanks of water of equal capacity.
One is full of cold liquid whereas another contains hot liquid.
What will happen if we put them to the frosting camera?
That’s right, while the cold tank will harden, the hot liquid will take some time to cool down first and only then it will begin to approach the frozen condition. Notoriously, the forming of ice begins at zero degrees Celsius, so the hot liquid will have to reach that point first before it can become solid. It may take twice as much time.

Also, it could be useful to refresh several most common questions asked regarding this point.
- How long does it take for cold water to freeze?
Normally, it will grow absolutely frozen after a maximum of four hours.
- How long does it take for hot water to freeze?
Since hot liquid requires more time to harden as it needs to cool down first, its time required for hardening will be something like twice as long as for cool liquid.
- How long does it take room temperature water to freeze?
Everything hangs upon the capacity of water, however, the average term is one and a half hours.
How Long Does It Take to Freeze Water?

It may seem that exposing water to frost is not a big deal but thinking like that isn’t quite correct.
This process is quite complicated by its nature and hangs upon different factors that influence the outcome greatly.
To manage to count the period required for some water to become solid, take into account the next nuances.
- The time required for complete freezing is highly dependent on water capacity.
- In addition, the initial temperature matters because hot or even warm liquid will need more time in comparison with the room temperature or cold liquid.
- The freezer output also matters.
- And, what may surprise, the tank plays a huge role, too! If the liquid is exposed to frost in a plastic tank, it will require more time to harden completely than in a metal tank.
And, what temperature does water freeze at in celsius? Water freeze at a temperature below 0° Celsius. Hot water freezes faster than cold water this is called Mpemba Effect. If the water isn’t pure, it will freeze at -2° or -3° degrees Celsius.
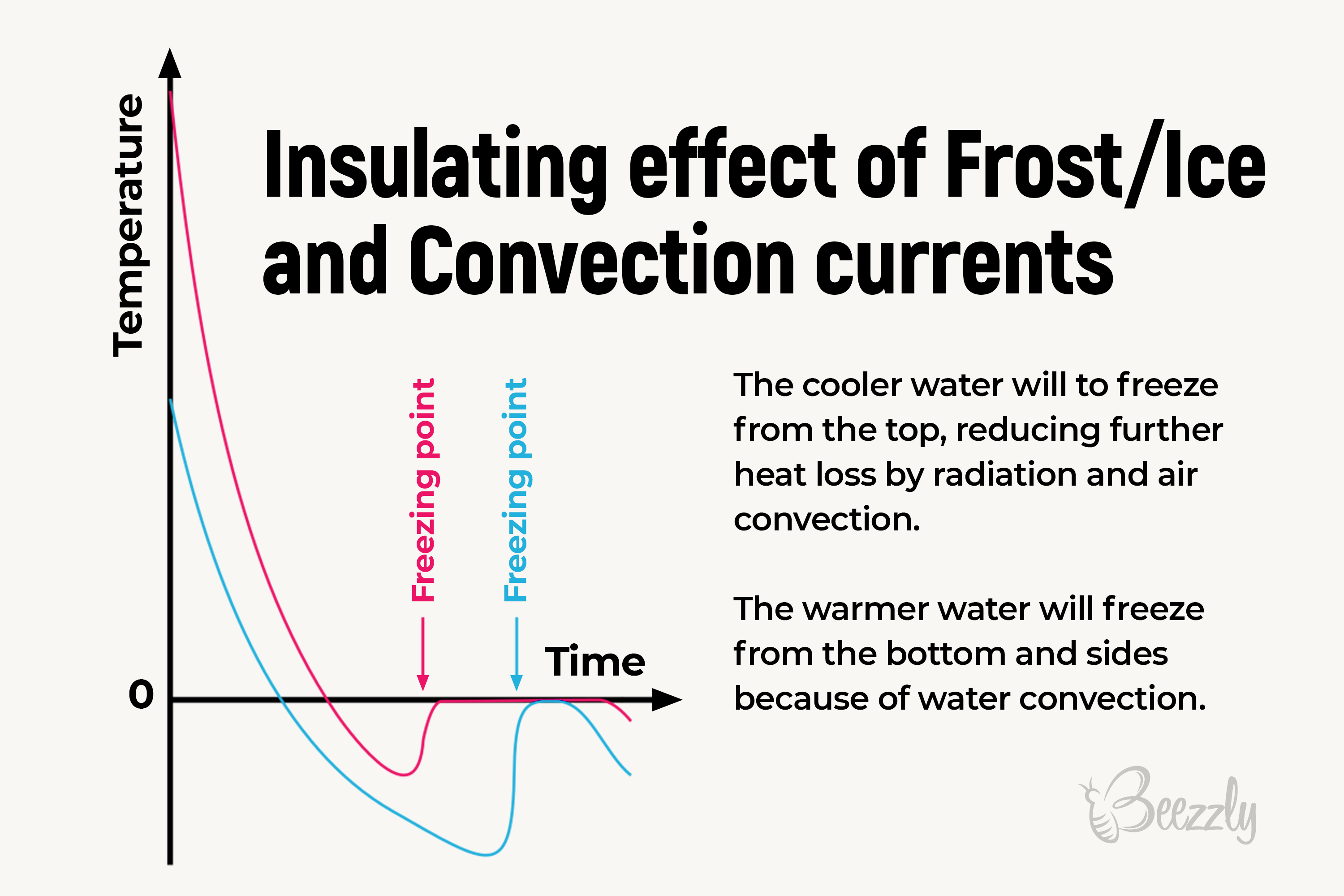
Does Water Hardens Equally Fast Everywhere?

Has anyone ever wondered whether the water hardens equally quickly in different surroundings?
Is water freezing time equal for the fridge frosting camera and the open-air pool like a lake, for instance?
In fact, the outcome differentiates. Since in the frosting camera, the liquid is affected by frost under the stable conditions, the open-air pools are affected by the constantly changing weather conditions and other fluctuations. This is the cause of why the “wild water” won’t freeze equally fast as the one we have at home.
How long does it take for water to freeze in the freezer?
Normally, it’s possible to get hard ice after four hours of instant frost exposure.
So, what temperature does water freeze in Fahrenheit? Water freeze at a temperature below 32° Fahrenheit. Hot water freezes faster than cold water this is called Mpemba Effect. If the water isn’t pure, it will freeze at – 35° or -38° degrees Fahrenheit.
Freezing Time For Different Capacity Of Water
To compare and realize the difference better, let’s verify the duration of the frost exposure process for different capacities of liquid.
- How long does it take for a cup of water to freeze?

One two hundred milliliters tank of liquid will require nearly three hours before hardening completely.
- How long does it take to freeze a gallon of water?
Using a powerful freezer, one gallon will most likely freeze after staying there overnight. However, other conditions must be also considered.
How long does it take to freeze a bottle of water?

From half an hour to two hours in general. Everything hangs upon the capacity of the bottle and the temperature in the freezer.
A one-liter tank will harden after two hours.
As we all know, water solidifies at 32 degrees Fahrenheit.
- So if we fill a half-liter tank with it and place it to the frosting camera with the ideal temperature, we can be sure we get it all-ice in thirty minutes.
- However, a one-liter bottle will take more time and, if being placed into a freezer with the proper level of cold, it will turn into ice after two hours of non-stop freezing.
Respectively, one can calculate the freezing time for a three-liter bottle and further up to as a voluminous tank as you need.
But be aware that these numbers are only valid for the ordinary home frosting cameras!
Colder freezers will make the liquid solidify faster hanging upon the temperature rate inside them.
How long does it take ice cubes to freeze?
From three to four hours. Since the majority of the ice-making trays that we use have a dozen of separate sections, they will freeze in a usual home frosting camera in about 3 hours. If you use a nonstandard tray – about 4 hours.
A conventional tray meant for dozen of ice pieces will become completely frozen in four hours at most.
Nevertheless, we would recommend everyone to consider certain nuances regarding freezing.
As everyone knows, the water turns into ice when it reaches the mark of zero degrees Celsius.
But how soon it will happen hangs greatly upon several aspects.
- The size of the cubes. If your tray is meant for making smaller ones, they will solidify way faster than those that are of a standard size or bigger.
- Consider the size of the surface of ice. Liquid turns into ice from the outside in. It means that if your tray has gaps filled with air between each cube, they will solidify much faster compared to those that only have plastic dividers between them.
- How cold is your frosting camera? The optimal level of cold is zero degrees Celsius, but if the camera was left open for a while or some warm foodstuff was placed in there, the balance will be disturbed, and the cubes will harden slower.
Do All Liquids Get Hard At the Same Temperature and the Same Fast?

Definitely, different kinds of liquids harden at a different temperature rate and under different conditions.
Everything hangs upon the specifics of the structure of each particular liquid.
The thicker it is the slower it will harden when frozen.
Respectively, the thinner it is the quicker the process will go.
Now the secret is revealed! So whenever some liquid needs to be transformed into ice, you will know for sure how much time to wait until the desired result will be received.
Interesting comment from the scientist Sarah Zelinski we get.
[expert_review params=”JTdCJTIyY29sb3IlMjIlM0ElMjJwdXJwbGUtMSUyMiUyQyUyMmV4cGVydF9hdmF0YXIlMjIlM0ElMjJodHRwcyUzQSUyRiUyRnRodW1icy1wcm9kLnNpLWNkbi5jb20lMkZiQzdMZXRNWjlNMGR0VGwxZFhydXMxRlE0aEklM0QlMkZmaXQtaW4lMkYxNjB4ODAlMkZmaWx0ZXJzJTNBbm9fdXBzY2FsZSgpJTJGaHR0cHMlM0ElMkYlMkZwdWJsaWMtbWVkaWEuc2ktY2RuLmNvbSUyRmFjY291bnRzJTJGaGVhZHNob3QlMkZTYXJhaC1aaWVsaW5za2ktMjQwLmpwZyUyMiUyQyUyMmV4cGVydF9uYW1lJTIyJTNBJTIyU2FyYWglMjBaaWVsaW5za2klMjIlMkMlMjJleHBlcnRfbGluayUyMiUzQSUyMmh0dHBzJTNBJTJGJTJGd3d3LnNtaXRoc29uaWFubWFnLmNvbSUyRnNjaWVuY2UtbmF0dXJlJTJGYXQtd2hhdC10ZW1wZXJhdHVyZS1kb2VzLXdhdGVyLWZyZWV6ZS0xMTIwODEzJTJGJTIyJTJDJTIyZXhwZXJ0X2lkJTIyJTNBJTIyJTIyJTJDJTIyZXhwZXJ0X3R5cGUlMjIlM0ElMjJzZWxmJTIyJTJDJTIyZXhwZXJ0X2F2YXRhcl9hbHQlMjIlM0ElMjIlMjIlMkMlMjJleHBlcnRfc2hvdyUyMiUzQTElMkMlMjJleHBlcnRfZGVzY3JpcHRpb24lMjIlM0ElMjJTYXJhaCUyMFppZWxpbnNraSUyMGlzJTIwYW4lMjBhd2FyZC13aW5uaW5nJTIwc2NpZW5jZSUyMHdyaXRlciUyMGFuZCUyMGVkaXRvci4lMjBTaGUlMjBpcyUyMGElMjBjb250cmlidXRpbmclMjB3cml0ZXIlMjBpbiUyMHNjaWVuY2UlMjBmb3IlMjBTbWl0aHNvbmlhbi5jb20lMjAlMjIlMkMlMjJleHBlcnRfc2hvd19idXR0b24lMjIlM0EwJTJDJTIyZXhwZXJ0X3Nob3dfYnV0dG9uX3R5cGUlMjIlM0ElMjJwb3B1cCUyMiUyQyUyMmV4cGVydF9xdWVzdGlvbl9idXR0b25fdGV4dCUyMiUzQSUyMkFzayUyMFF1ZXN0aW9uJTIyJTJDJTIycXVlc3Rpb25fZXh0ZXJuYWxfbGluayUyMiUzQSUyMiUyMiUyQyUyMnBvcHVwX3VzZV9waG9uZSUyMiUzQTAlMkMlMjJleHBlcnRfdGl0bGUlMjIlM0ElMjJXYXRlciUyMGRvZXNuJUUyJTgwJTk5dCUyMGFsd2F5cyUyMHR1cm4lMjB0byUyMGljZSUyMGF0JTIwMzIlMjBkZWdyZWVzJTIwRmFocmVuaGVpdCUyMiUyQyUyMmV4cGVydF9zaG93X3RpdGxlJTIyJTNBMSUyQyUyMmV4cGVydF90ZXh0JTIyJTNBJTIyV2UlRTIlODAlOTl2ZSUyMGFsbCUyMGJlZW4lMjB0YXVnaHQlMjB0aGF0JTIwd2F0ZXIlMjBmcmVlemVzJTIwYXQlMjAzMiUyMGRlZ3JlZXMlMjBGYWhyZW5oZWl0JTJDJTIwMCUyMGRlZ3JlZXMlMjBDZWxzaXVzJTJDJTIwMjczLjE1JTIwS2VsdmluLiUyMFRoYXQlRTIlODAlOTlzJTIwbm90JTIwYWx3YXlzJTIwdGhlJTIwY2FzZSUyQyUyMHRob3VnaC4lMjBTY2llbnRpc3RzJTIwaGF2ZSUyMGZvdW5kJTIwbGlxdWlkJTIwd2F0ZXIlMjBhcyUyMGNvbGQlMjBhcyUyMC00MCUyMGRlZ3JlZXMlMjBGJTIwaW4lMjBjbG91ZHMlMjBhbmQlMjBldmVuJTIwY29vbGVkJTIwd2F0ZXIlMjBkb3duJTIwdG8lMjAtNDIlMjBkZWdyZWVzJTIwRiUyMGluJTIwdGhlJTIwbGFiLiUyMCU1Q24lMjIlMkMlMjJxYSUyMiUzQSU1QiU1RCUyQyUyMnFhX3Nob3dfdGl0bGUlMjIlM0ExJTJDJTIycWFfdGl0bGUlMjIlM0ElMjJRdWVzdGlvbnMlMjB0byUyMHRoZSUyMGV4cGVydCUyMiUyQyUyMnNjb3JlJTIyJTNBJTVCJTVEJTJDJTIyc2NvcmVfc3VtbWFyeV90ZXh0JTIyJTNBJTIyJTIyJTJDJTIyc2NvcmVfc3VtbWFyeV9hdmVyYWdlJTIyJTNBMCUyQyUyMnNjb3JlX21heCUyMiUzQSUyMjUlMjIlMkMlMjJzY29yZV9zeW1ib2wlMjIlM0ElMjIlMjIlMkMlMjJzY29yZV90aXRsZSUyMiUzQSUyMiUyMiUyQyUyMnNjb3JlX3Nob3dfdGl0bGUlMjIlM0EwJTJDJTIycGx1c2VzJTIyJTNBJTVCJTVEJTJDJTIybWludXNlcyUyMiUzQSU1QiU1RCUyQyUyMnBsdXNlc19taW51c2VzX3Nob3dfdGl0bGUlMjIlM0ExJTJDJTIycGx1c2VzX21pbnVzZXNfdGl0bGUlMjIlM0ElMjJQbHVzJTIwJTI2JTIwTWludXMlMjIlMkMlMjJwbHVzZXNfdGl0bGUlMjIlM0ElMjIlMjIlMkMlMjJtaW51c2VzX3RpdGxlJTIyJTNBJTIyJTIyJTdE”]
The Abnormal Properties Of H2O

Have you ever thought of those mysterious and unexplained traits of such a common liquid as water?
This substance has become so familiar for us that we don’t even guess how many curious facts about it we don’t even know.
Shall we dive into the world or mystery together?
- Are you aware that aqua solidifies from the surface to the bottom? If it was vise versa, all the fish in our lakes would not survive the first frosts!
- Liquid aqua is heavier than its hard icy counterpart
- Aqua has the ability to save heat very well, thus lakes and ponds don’t freeze so fast
- H2O is a great solvent. Due to its polarity, this liquid can dissolve surprisingly effectively
- It has the highest surface tension of all known liquids!
- Since H2O molecules have polarity, the liquid has a high cohesion (the force that holds the molecules together). It makes H2O molecules hold together even in a single drop, and that’s the reason why we can fill a tank with this liquid above the rim and it won’t spill!
- Water can transmit electricity, that is why it is prohibited to touch the wires and plugs with the wet hands or spill aqua on the working electricity devices
- Recent researches showed that H2O molecules react to the words and other sound vibrations made nearby. This liquid literally “listens” to the sounds from outside and responds by changing its molecular structure!
As you can see, H2O is indeed a tricky and mysterious substance that still has a lot of secrets that our science is only about to discover!
Frequently Asked Questions About How Water Freezes
Freezing water can hardly be called something that common people are interested in.
Nevertheless, when we need to chill something liquid at home, this issue becomes a true point of concern!
That is why so many of us have lots of related questions regarding different issues connected with freezing water and other liquids.
To help you out to answer those questions and to make it simpler to sort out the facts, we have collected the most common questions asked by people, each with the detailed answer.
We hope you will find the information we provide you with useful!
How long does it take to make ice cubes?

Quite a widespread belief exists that the solidifying period needed for water in an ice cube form to harden completely hangs upon the number of sections this form consists of.
In point of fact, what really matters is the amount of substance in each section.
No matter how many sections the tray has, the average period needed until the substance freezes is usually three to four hours.
Nevertheless, if your particular tray has deeper (or shallower) sections, the amount of time might be less extended.
How long does ice take to freeze on a lake?
Sometimes knowing how fast ice forms can be even life-saving, especially when it comes to wild nature aqua, for instance, lakes or rivers.
In such a case, the answer hangs upon the size of the basin, depth, plus the air temperature around.
Everyone is informed that water freezes at 32 F but it doesn’t necessarily mean the pond or lake surface will become solid ice once the air temperature reaches that point!
According to science, since water is very good at holding heat, the more sizeable the lake/pond is the slower its water molecules will create the ice.
For this reason, a large lake freezes after at least a week. Besides, in view of this we can often see those melted wholes on the basin’s frozen surface.
The science researches show that normally an ice film on the surface of such a basin grows for 1 inch per each fifteen freezing degree days in a twenty-four hours period.

How long does it take for water to freeze in a freezer?
Sometimes it is useful to know how soon we can get our ice cubes, or some ice-cold water, right?
Well, the answer to this inquiry hangs upon the amount of liquid we chill.
Primarily, if we place a 200 ml flask with aqua to the frosting camera, it will change into ice after approximately three hours.
In contrast, the content of an ice tray will harden after the four hours of contact with frost.
Moreover, if we expose water to heat, it will turn into ice faster!
How long does it take water to freeze at 32 degrees?

Basically, the answer says: the more sizeable the tank is the longer it needs to shapeshift into ice. However, the level of heat the liquid had before it was placed to the frosting camera also matters.
As everyone knows, water freezes best at 32 F. So normally, a small portion of the aqua freezes after three to four hours spent in the frosting camera of the fridge.
However, the effect will hang upon one more factor which is the purity of water. According to science, pure water (like distilled or filtered) hardens the best.
At what temperature does pure water freeze? The crystal clear water will freeze at a temperature below 32° Fahrenheit. If the water isn’t pure or if you are in the mountains, the freezing point water will increase up to 35° – 45° Fahrenheit.
How long does it take a water bottle to freeze?
The answer to this inquiry will hang upon the capacity of the flask. Usually, one liter of H2O changes into ice after two hours of chilling.
How long does it take for the juice to freeze?
Some people think that juice since being aqueous just like H2O chills the same fast.
Unfortunately, it is time to upset you guys since fruit juice (just like any other type of this drink, e.g. berry or veggie juice) requires more time in the frosting camera to have any significant effect!
The least interval needed for a packet of fruit beverage to solidify is nearly six hours so the best thing one is able to make to shapeshift the juice into ice is to toss it to the frosting camera overnight.

Does boiled water freeze faster?
Our science says that not all aqua solidifies equally. Even though water molecules react to frost the same way, the final effect will appear after a different period of time.
If the liquid was initially pure (for instance, if we used distilled or filtered water), its surface will freeze best compared to the water taken right from the tap.
However, the most curious thing about this issue is that simmered or just hot water tends to harden quicker than the cold one!
Science hasn’t discovered the proper reply to this inquiry yet, besides, no data exists that can describe what this effect is related to.
What liquid freezes the fastest?

Science states that aqua is the only liquid known on Earth that turns into ice faster than all the other liquids on our planet.
The way science explains this phenomenon includes the statement that water is an originally pure liquid since it doesn’t contain any additives or other components. It is only hydrogen and oxygen.
Thus, after the contact with frost, it hardens so quickly and easily.
Also, science discovered that, after the impact of heat, simmered H2O shapeshifts into ice quicker than its cold counterpart.
Water Freezing Surprising Facts. We’ll Answer All Questions Of Yours

What is so mysterious and unknown about freezing water, you might ask?
Well, indeed, such a common effect that cold and frost has on the most widespread liquid on Earth seems to be already explained by the science.
Nevertheless, people keep on asking an enormous amount of questions about related topics such as the highest/lowest temperature water freezes at, or how exactly it can form itself into ice.
Sometimes it even seems that those inquiries appear faster than science develops!
Of course, it is impossible to find an answer to every single inquiry but we decided to do our best and provide you with those that we can find out about.
Want to know how water freezes? Keep on reading and you’ll get closer to science and find the answer to the related questions.
- What is the lowest temperature water freezes at?

That’s an interesting question! We have been told since childhood that water freezes and turns into ice at zero degrees. However, science as proved quite the opposite! It has been revealed that pure liquid water freezes at a much lower degree of -38 C becoming super cool!
How is this effect explained? The answer will surprise everyone! For water to form ice at the temperature higher than -20C, it needs a solid particle called nucleator.
This particle will initiate water molecules to nucleate and form ice crystals on the surface.
So the science investigations found out that dissolving certain substances in water can change the speed of the nucleation process.
The second these elements contact water, they can either speed up or slow down the water molecules to freeze and change into a solid form.
For instance, the contact with the sea salt makes ice-forming slower whereas ammonium sulfate (it’s also salt) has an opposite effect.
- How fast can water evaporate?
Of course, the correct answer would be that it hangs upon the amount of liquid and the intensity of the heat. The science researches found out that such amount of liquid as one standard cup of water will fully evaporate after 1.2 hours of constant boiling at the same heat level.
- Why does water evaporate faster when increasing heat?
The answer is the following. Moving water evaporates faster than still water. It means that when we increase the heat, the molecules start moving faster rubbing against each other which warms the water up after a while. So if we change the heat and make it higher, our liquid will boil sooner. Also, this is the answer to the question of why water rolls while boiling.
- In what sort of container is it best to freeze water?

As we all know, when water freezes it expands. Science has proved that a long time ago. That is why for freezing this liquid it is best of all to not choose hard tanks or porcelain vessels. Solidified water can easily damage the surface of the container and even crack it. So, tossing your fitness bottle with water to freeze it is definitely not the best idea!
- Why do I always find air bubbles in ice cubes?
Science is a wonderful thing! You probably asked yourself many times why you find air bubbles packed under the surface inside of the ice cubes, right? In fact, the explanation is simple.
The air that is thrown out from the freezing surface becomes erupted right into the ice cube when water freezes. That is why we often find those floating bubbles inside when taking the ice tray out of the frosting camera.
How long does it take for the ground to freeze?
Ground freezing takes from 6 to 8 weeks at 32° Fahrenheit or below 0° Celsius, at small gardens, and from 10- to 12 weeks on the fields. And it will be frozen until the temperature gets 40° Fahrenheit or + 4° Celsius.
And, how long does it take for snow to melt at 35 degrees? Usually around 5 hours. But it depends on the amount of snow, the thickness of the ice, and is it rainy outside or not. An ordinary 6 inches of snow would melt in about 5 hours.

Does hot water freeze faster than cold water? I heard it is possible but could someone explain why that happens?
Yeah, it is in fact real! Hot water solidifies faster than its cold counterpart! Even scientists are still surprised. As far as I remember, it was explained that when hot water cools down, it loses its mass to evaporation. Having less mass, the liquid must lose less heat to chill, this is why it freezes quicker.
You say early on in this that ice is denser than water – it isn’t, water is more dense, and unusual in that it is less dense in its frozen and gas form that its liquid state – also ice floats; if it were more dense than water it would sink to the bottom 🙂
Hello! Thank you for your response! We have double-checked your clarification regarding the water/ice density issue, and yes, we have to admit that you’re right) Frozen water is less dense than the liquid one even though ice may seem heavier by sight. Thanks for being such an attentive reader! Regards, Manager Olivia
Hi! Is there anyone here who is familiar with piping more or less? Will pipes freeze at 32 degrees? I live in a quite cold area and I need to decide whether to extra-protect my pipes or not. Thanks!
Hey there! Well, that’s an arguable question. Water does indeed freeze at 32F. But the pipes must already be protected, aren’t they? I mean that even those we have in the attic or in the garage have some protection from cold and frost.
Anyway, according to my experience, the outdoor temperature must be at least 20F to make pipes freeze.
How long does it take water to freeze at 32? I found various answers on internet but maybe someone has done it themselves to check? I’m so curious to figure this out! Thanks!
Well, if we take an ordinary home freezer and an ice-making tray of a standard size, the water in it will solidify in approximately four hours. As you could guess, everything depends on the amount of liquid.
I’ve been always wondering: will saltwater freeze in a freezer?
I guess it won’t freeze. Salty water will solidify at -21C or colder. Since our home freezers have only -18C, it’s impossible to freeze this kind of liquid in them.
Sorry if I disappointed you!
Does freezing water purify it? Are there any other methods for purifying water?
Freezing can indeed help to make the water cleaner. When it freezes slowly, the impurities are forced up, and there is no room left for contaminants.
Other methods are distilling, boiling, filtering, and using chemicals.
What freezes water instantly? Just curious!
As far as I remember it from our chemistry lessons, dry ice and ethanol mixed together freeze water immediately. Also, liquid nitrogen works well. Maybe there are some other ways, I don’t know.
Will water freeze at 33 degrees or when it’s colder?
Yes, water can solidify at 33 or lower. In fact, scientists even found water being frozen at -40 F in the clouds.
How can you make water freeze faster? I’m always bored to wait until that ice freezes in the ice tray!
Hey! I’d recommend you go for a metal ice tray, copper, for instance. Metal will remove the warmth from the water faster thus it’ll turn into ice quicker.
Of at least try to chill the empty tray in the freezer before pouring water in it.
Hi everyone! Maybe, someone can help me to solve this mystery: when water freezes and turns into ice, it’s of 32 F. But what happens when ice keeps on staying frozen? Can ice be colder than 32?
Hey! I guess it can, at least, I read somewhere that it is possible to make ice colder than 32 F. But I’m pretty sure it can be done only in a lab!
How long does it take for 4 inches of water to freeze? Thanks in advance for your answers!
I was once making an experiment and froze 8 inches of water in 72 hours at -10 C. So if you have the same conditions, you’ll freeze 4 inches in 36 hours.
I noticed that water bottles in my garage start freezing slowly. It’s pretty cold in there since I’m not using the garage and it’s not warmed. Do you think they can freeze completely? How long does it take for 500ml of water to freeze?
It depends on the temperature in your garage. I used to freeze a 500 ml bottle of water completely at -12C, but that was outside.
Please help me to figure out one issue: are 32 degrees above freezing?
No, 32 F is the freezing point actually.
hi
Can water freeze in 2 hours? See, I need to make ice cubes for the home party. The party is tomorrow evening, and the freezer is already stuffed, so I don’t have that much time left for the cubes to freeze. Do you think I’ll manage for a couple of hours tomorrow?
I’m pretty sure you will if the cubes are small. Home freezers usually take up to 2 hours to freeze water and other liquids, so there should be no problem. But I’d still recommend you place the cube tray into the freezer in advance so that they have an extra hour just in case.
Does hot water freeze faster than cold water?
I’m pretty sure the answer is yes. I remember I once saw a TV science series where they showed how hot water freezes faster than cold water. This phenomenon is surprising even to most scientists, but it is in fact real. It has been seen and studied in numerous experiments.
How long does it take for water to freeze into ice cubes? I’m getting ready for the party and we need a lot of ice cubes, but I need to know how much in advance I should start making them!
Hi! Well, usually, in a regular home freezer, ice cubes will freeze solid in a couple of hours. But if your freezer is stuffed with food or bottles with water, this may take more time!
Thanks for explaining how much time it takes water to freeze. But I have another question. Will water freeze at 32 degrees?
Hello. The rule of thumb is that water freezes at 32 degrees Fahrenheit (0 degrees Celsius), but it can actually stay liquid over a range of chilly temperatures. It depends on certain conditions.